Showing 11 – 20 of 122 results Showing all 122 results Showing the single result No results found
Category
Filters Sort results
Reset Apply
Preview | Product Name | Composition | Division |
|---|---|---|---|
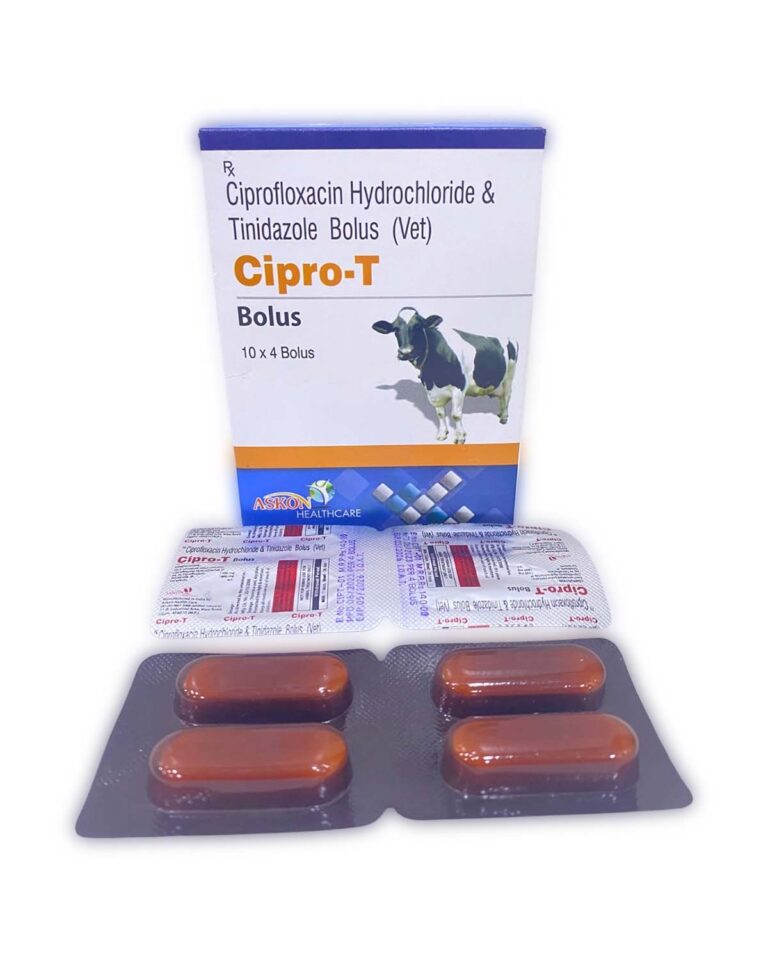
CIPROFLOXACIN HYDROCHLORIDE IP eq. CIPROFLOXACIN IP 1500mg ⋅ TINIDAZOLE IP 1800mg | Veterinary | ||
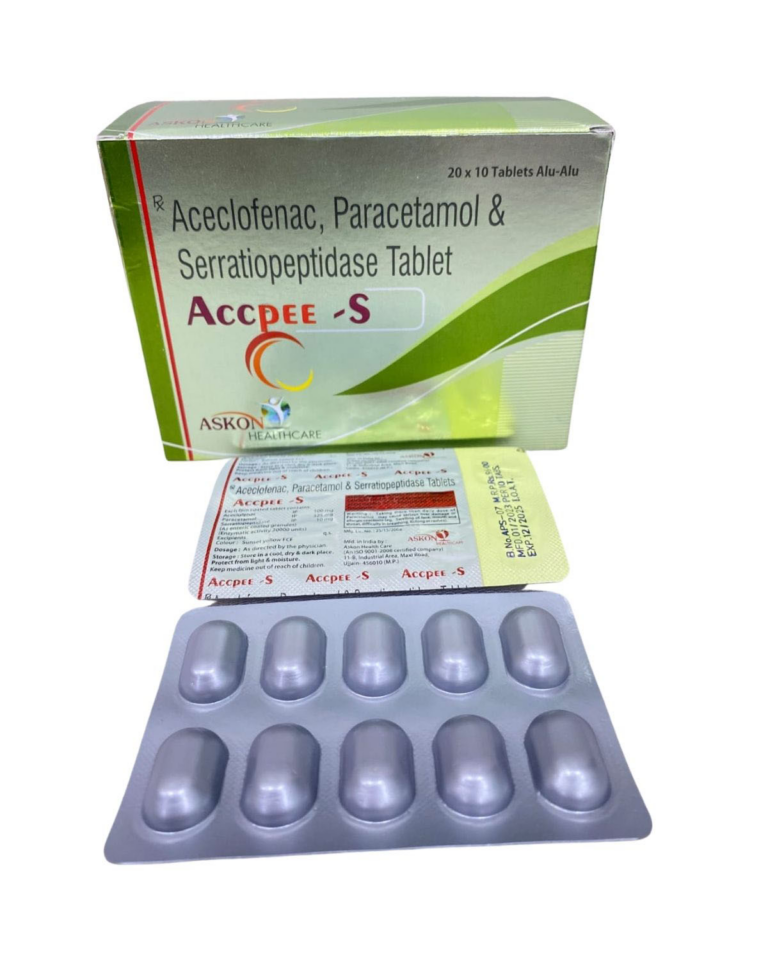
ACECLOFENAC IP 100mg ⋅ PARACETAMOL IP 325mg ⋅ SERRATIOPEPTIDASE IP 10mg | Domestic | ||
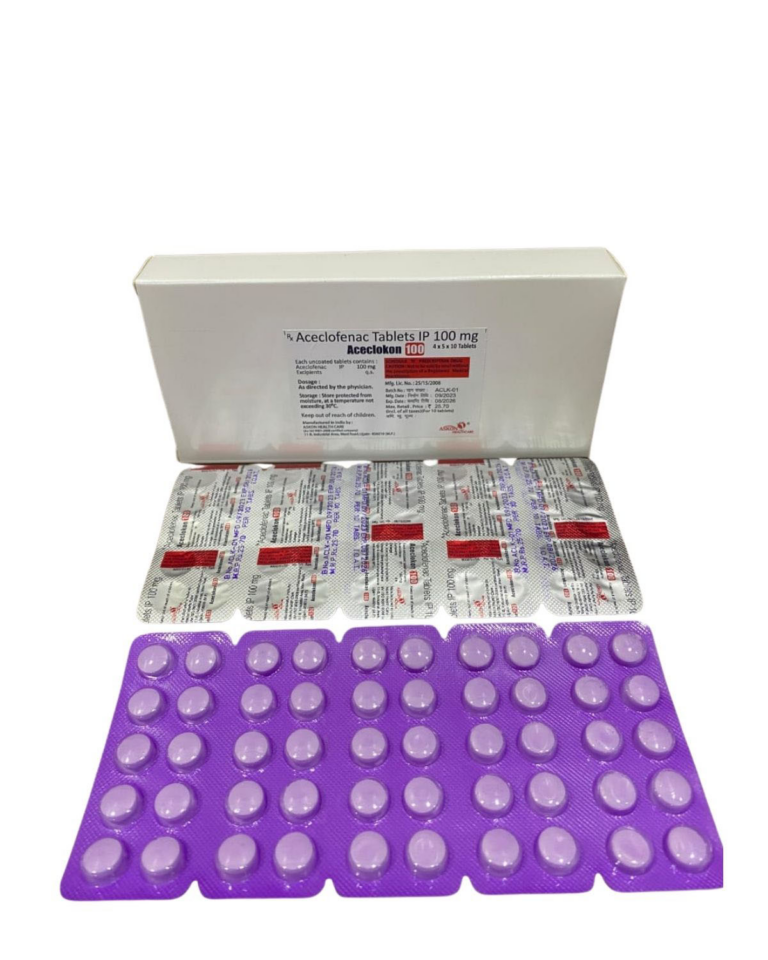
ACECLOFENAC IP 100mg | Domestic | ||
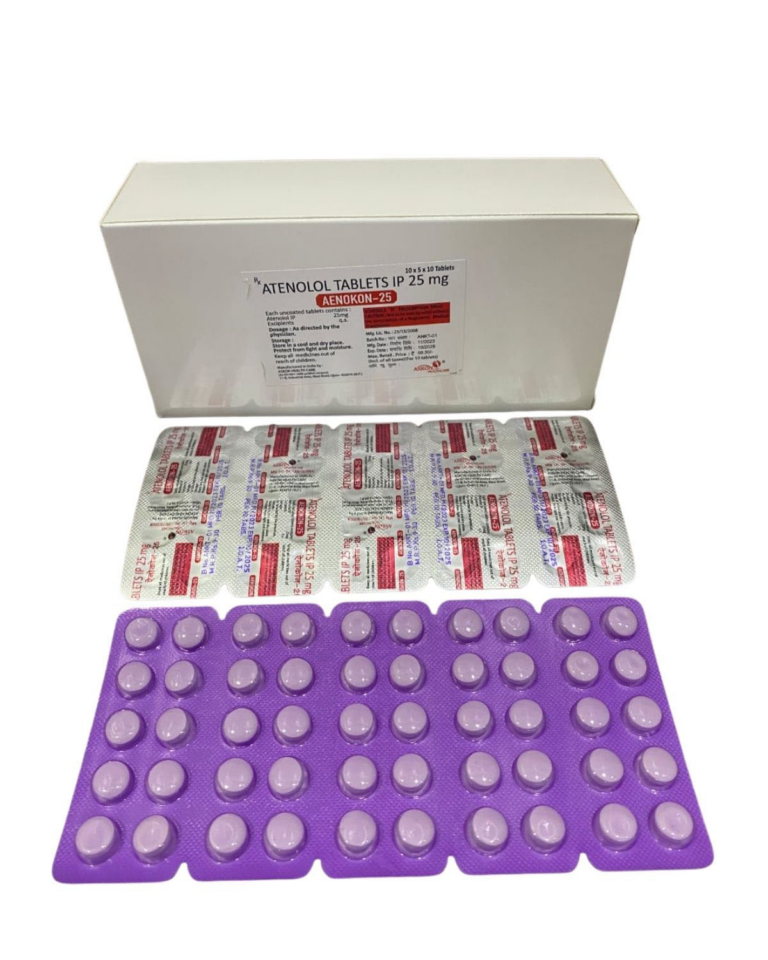
ATENOLOL IP 25mg ⋅ ATENOLOL IP 50 MG | Domestic | ||
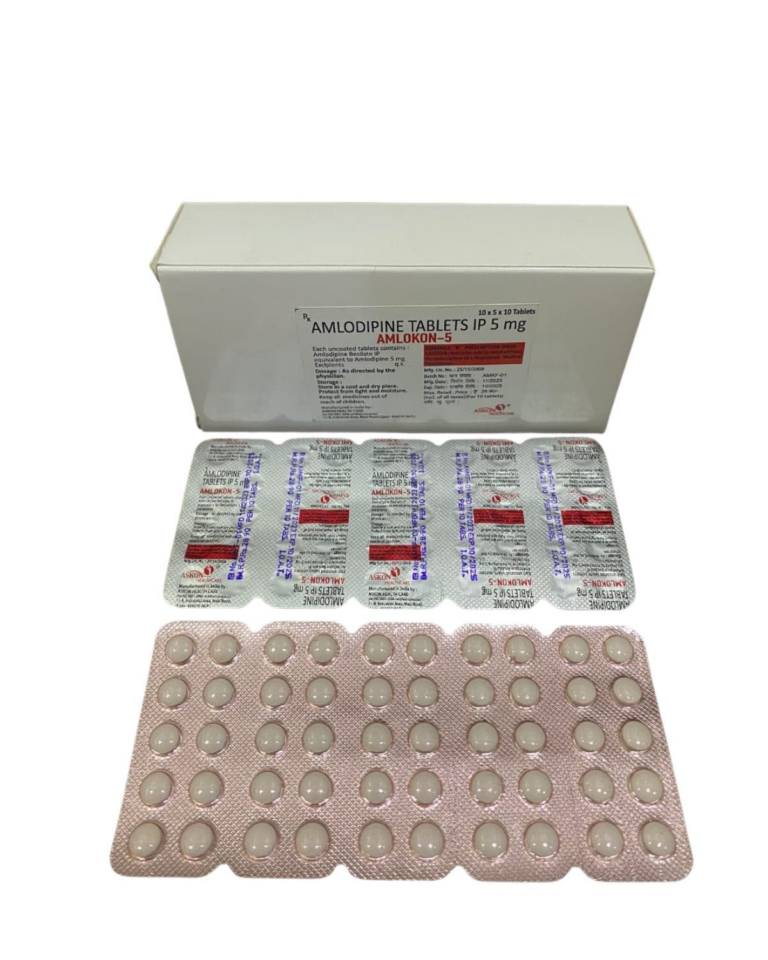
AMLODIPINE BESILATE IP EQ. TO AMLODIPINE 10mg ⋅ AMLODIPINE BESILATE IP EQ. TO AMLODIPINE 5mg | Domestic | ||
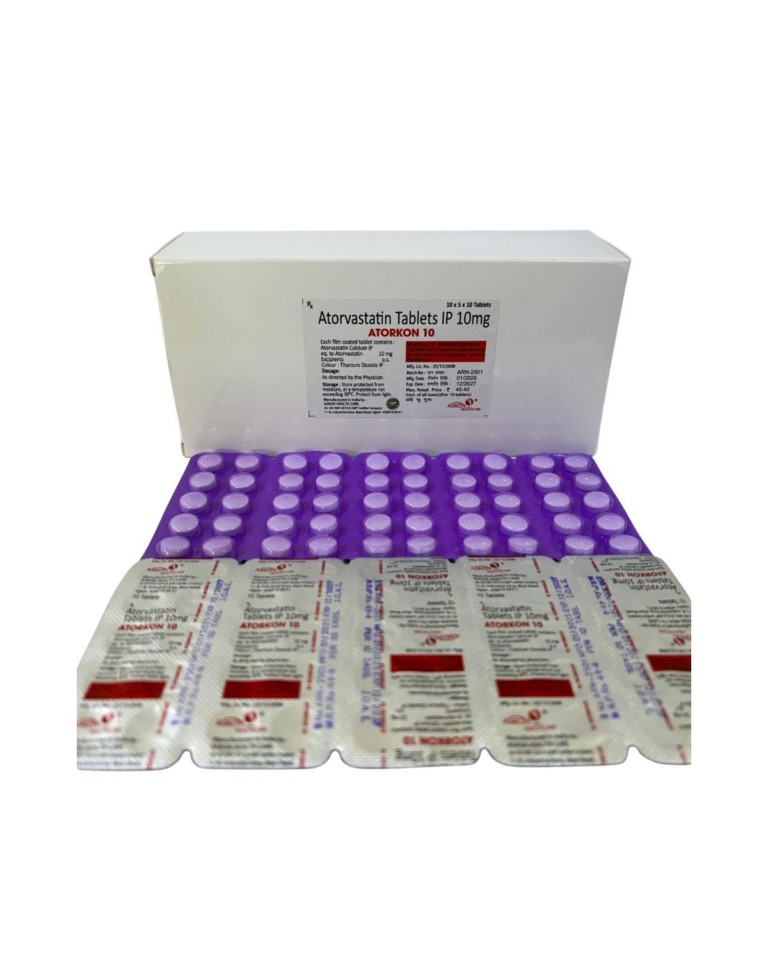
ATORVASTATIN CALCIUM IP EQ. TO ATORVASTATIN 10mg ⋅ ATORVASTATIN CALCIUM IP EQ. TO ATORVASTATIN 20mg | Domestic | ||
DICYCLOMINE HYDROCHLORIDE IP 10mg ⋅ DICYCLOMINE HYDROCHLORIDE IP 20mg | Domestic | ||
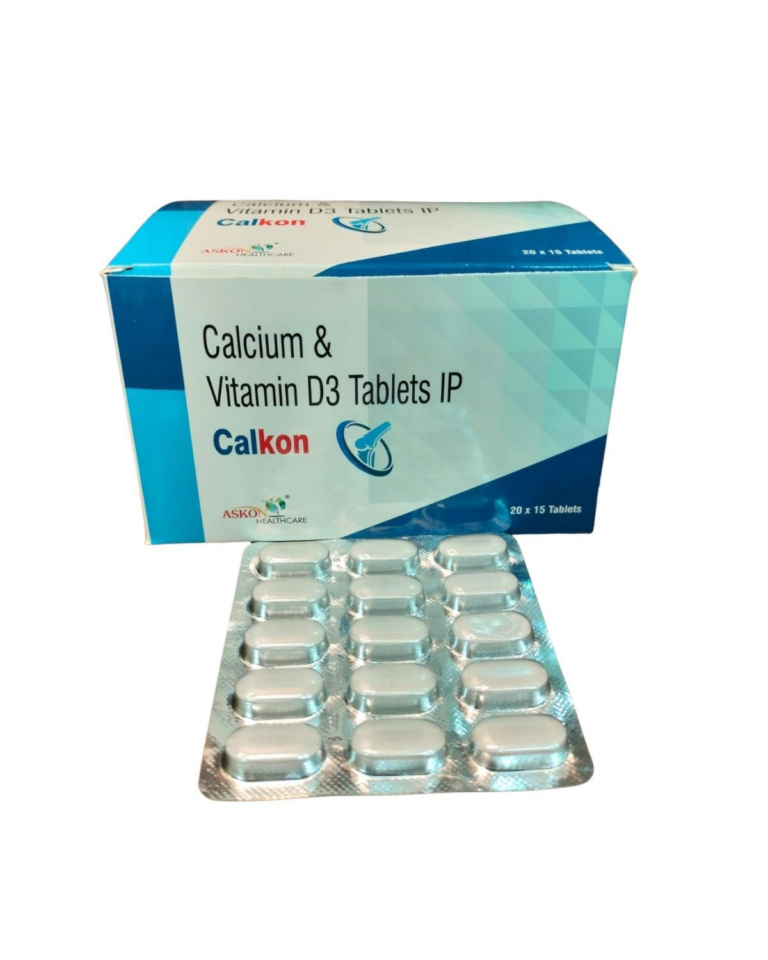
CALCIUM CARBONATE IP 1.25mg EQ. TO ELEMENTAL CALCIUM 500mg ⋅ CHOLECALCIFEROL (VITAMIN D3) IP 250mg | Domestic | ||
CETRIZINE HYDROCHLORIDE IP 10mg | Domestic | ||
CHLORPHENIRAMINE MALEATE IP 4mg | Domestic |
[Product_Table id='4835' name='Product Archives']
Manufacturing That Defines ASKON
Excellence That Inspires Trust
Our manufacturing facilities are built to meet the most stringent Healthcare standards, equipped with modern technology and automated systems for precision and hygiene. Every product is processed in controlled environments, ensuring consistency, purity, and efficacy. From sourcing raw materials to final packaging, each stage is carefully monitored by qualified professionals — supported by ISO and GMP-certified processes that uphold our dedication to quality, safety, and global compliance.
01
GMP & ISO 9001:2015 Certified Units
02
Automated Production & Packaging Lines
03
Strict Quality Control at Every Stage
04
Cleanroom & Environmental Safety
[elementor-template id="2622"]